Do You Read an Elisa With the Stop Solution
Practice you Oft Trouble in These Problems of ELISA?
Enzyme-linked immunosorbent assay, too known as ELISA or Environmental impact assessment, is a plate-based assay technique designed for detecting and quantifying substances such every bit peptides, proteins, antibodies and hormones by changing antibodies and color to identify a substance. In an ELISA, an antigen must exist immobilized on a solid surface and so combined with an antibody that is linked to an enzyme. Detection is accomplished past assessing the conjugated enzyme activity via incubation with a substrate to produce a measureable production. The most crucial element of the detection strategy is a highly specific antibody-antigen interaction. Information technology is a common test that detects and measure out antibodies in your claret, so that an ELISA test may be used to diagnose some diseases, such as HIV-a cause of AIDS, Lyme illness, pernicious anemia, Rocky Mount spotted fever, rotavirus squamous cell carcinoma, syphilis, et al.
CUSABIO has a sound platform for the development of ELISA kit, mature antigen-antibiotic research and development system. We are skillful in a diversity of ELISA technologies, such as the double-antibody sandwich method, double antigen sandwich method, (direct) competition ELISA, indirect contest ELISA, blocking method, indirect ELISA and other methods. Combined with diagnostic kits development squad, we are able to develop the kits with clinical diagnostic level, and make the quality in the leading place worldwide. Currently, CUSABIO now offers a broad range of ELISA kits covering over 9,000 different assay targets.
In this commodity, nosotros primarily focus on the common bug of ELISA experiment.
ane. What is the common process of ELISA?
ELISA is the abbreviation of the enzyme-linked immunosorbent assay used to identify the presence of specific proteins and to determine their concentrations. In that location are iv types of ELISA including sandwich, competitive, indirect and direct ELISA. The common procedures are Storage, Reagents grooming, ELISA plate, Samples or reagents adding, Incubation, Washing and Reading. Please refer to our ELISA protocol for details. As shown in Figure 1, there are five procedures of ELISA in brief.
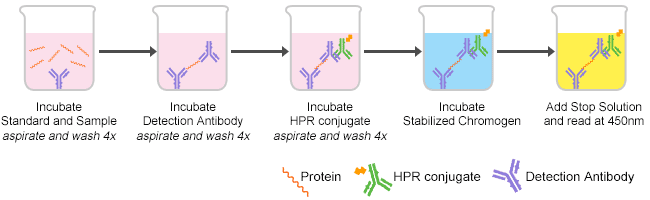
Figure one. The procedures of ELISA
2. Several bug of the color reaction in ELISA experiment
The ELISA results are generally based on the color depth of the chromogenic substrate. The color reaction is required to exist carried out at 37 °C for about 10 minutes, so the color reaction is terminated with a cease solution, and the absorbance at a specific wavelength is monitored with a microplate reader. Since the detection path of each reaction well is perpendicular to the microplate reader, the bottom of each reaction well should exist kept clean when testing, and the amount of chromogenic substrate and stop solution should be accurate, because the within of the well The amount of liquid volition affect the final caste. In this part, we summarize several results of the color reaction in ELISA experiment. Too that, every bit shown in Table 1, nosotros also conclude the causes and solution, respectively.
Tabular array 1. The bug of the color reaction
No indicate
| crusade | solution |
|---|---|
| Assay ready incorrectly, used incorrect reagents or Incorrect wavelength | Review protocol. Repeat assay using a positive control and check plate reader for wavelength, filters, gain etc. |
| Not enough antibiotic used | Try different concentration of the chief and/or secondary antibiotic. |
| Incubation fourth dimension too brusque | Incubate samples overnight at 4°C or follow the manufacturer guidelines. |
| Antibody stored at 4°C for several weeks or subjected to repeated freeze/thaw cycles | Employ a fresh aliquot of antibody that has been stored at -20°C or below. |
| Recognition of epitope impeded past adsorption to plate | To heighten detection of a peptide by direct or indirect ELISA, conjugate peptide to a large carrier protein before coating onto the microtiter plate. |
| Dull color evolution of enzymatic reaction | Prepare substrate solution immediately earlier use. Ensure the stock solution has not expired and is not contaminated. Permit longer incubation. |
Low sensitivity or weak betoken
| cause | solution |
|---|---|
| Improper storage of ELISA kit | Store all reagents equally recommended. Please note that all reagents may not have identical storage requirements. |
| Plate reader settings incorrect | Check plate reader for wavelength, filters, gain, etc. |
| Inactive detection reagent or detection reagent too dilute | Ensure reporter enzyme/flour has the expected activity, or use a college concentration of detection reagent. |
| Insufficient amount of antigen was coated to microtiter plate | Apply more antigen for blanket or very coating buffer. |
| Not enough antibody used | Increment concentration of the main and/or secondary antibiotic. Optimize antibody concentrations for your analysis. |
| Mixing or substituting reagents from different kits | Avoid mixing components from different kits. |
| Incubation temperature as well low | Optimize the incubation temperature for your analysis. Reagents should be at room temperature before beginning the assay. |
| Assay plates were compromised or previously used. | Be sure to air-condition plates in sealed bags with a desiccant to maintain stability. Prevent condensation from forming on plates by allowing them to equilibrate to room temperature while in the packaging. If partial plates are used, you must exist sure to characterization used wells to prevent reuse; cover them with sealing tape and utilize the remaining wells every bit soon as possible. Do non store partially used plates with other plates. Include a desiccant in the storage bag. |
Loftier groundwork signal
| cause | solution |
|---|---|
| Besides much antibody used | Optimize antibiotic concentrations for your analysis with unlike dilutions. |
| Too much detection reagent | Ensure the reagent has been diluted properly or repeat assay with a higher dilution of detection reagent. |
| Incubation temperature as well loftier | Attempt different temperature for optimizing your assay. |
| Reaction not stopped | Apply stop solution to foreclose overdevelopment. |
| Waiting too long to read plate after adding stop solution | Read plate immediately after adding end solution. |
| Incubation with substrate carried out in the low-cal | Perform substrate incubation in the dark. |
| Non-specific binding of antibiotic | Use a suitable blocking buffer or utilise an affinity-purified antibody. |
| Muddy plate | Clean the plate lesser advisedly and reread. |
Slow color evolution
| cause | solution |
|---|---|
| Incubation temperature is incorrect | Ensure plates and reagents are kept at room temperature. |
| Contaminated solutions | Make fresh solutions. |
| Detection reagent too old, contaminated or used at the wrong pH | Use fresh detection reagents at the correct pH. |
| Incorrect cohabit was used, conjugate was prepared incorrectly or has deteriorated. | Be sure that the conjugate used is the i that came with the kit. All conjugates are kit- and lot-specific. If preparation of a working conjugate is needed, be certain that the concentrate and diluent are mixed in correct volumes. Do non set up the working solution too far in accelerate and do not save whatsoever unused portion for future use. If no conjugate preparation is necessary, be sure to pour out only the amount required for immediate employ and do non return whatsoever unused portion to the stock canteen. |
| Wash buffer contains sodium azide. | Avoid sodium azide in the launder buffer. |
three. Other problems of ELISA experiment
In that location are numerous problem in ELISA experiment except for abnormal color reaction, involving standard bend, data analysis, the repeatability of experiment, et al. In this role, we list a few representative questions as follows, if you desire to know more than, delight click the below link: https://www.cusabio.com/yard-307.html.
Why does the standard curve look poorly?
In the ELISA data assay, the standard curve is the most important factor for tested samples. Therefore, how to brand a fine standard curve and the quality of the standard are critical. Here, nosotros conclude several reasons and solutions near the question that Why does the standard curve look poorly every bit follows. If yous want to obtain the manner of making standard bend, please click hither.
a. Standard solution is Improper, y'all should confirm dilutions and brand them correctly.
b. Standard is reconstituted improperly. Yous should spin vial briefly before opening and inspect for undissolved material subsequently reconstituting.
c. Standard is degraded, you should store and handle standard as recommended.
d. Curve doesn't fit scale, please try plotting using different scales e.g. log-log, five parameter logistic curve fit.
due east. Pipetting error, Using calibrated pipettes and proper pipetting technique to revise.
Why does it read low with normal results by eyes?
Wrong filter is used when taking readings. Wavelength should exist 450nm with a 650nm wavelength correction for TMB. Checking plate reader for wavelength, filters, proceeds etc. again before reading.
Why is repeatability poor?
a. Improper storage of the kit or poor storage environment. You should shop all components as recommended on data sheet rather than room temperature for excess time.
b. Incorrect preparation of standard. You should reconstitute standard strictly with the recommended diluent according to the kit protocol. You should prepare reagents in 10 minutes prior to use and add them to wells promptly.
c. Insufficient mixing after adding samples. Y'all should fully mix reagents in the vortex mixer when adding several reagents at the same time. Be conscientious when holding reagents to avoid splashing.
d. Poor repeatability of the plate readings. You lot should calibrate the plate reader.
e. Inconsistent incubation fourth dimension, washing condition, color development condition and operators. You should echo the assay of standard. Ensure consistent reactivity condition and operators.
f. Improper washing. You should add 200μL of wash buffer or fully make full into every well with pipette just no overflows are allowed. Bank check that all ports of the plate washer are unobstructed to ensure sufficient washing.
one thousand. Uneven temperature. You should proceed constant temperature during incubation to avert temperature fluctuations.
h. Too much remainder on the wall of the wells when calculation or the bottom of wells scratched with pipette tip. You should lower the pipette tips forth the wall of wells when adding slowly and carefully. Do not affect the bottom of wells.
i. Reused materials. You should modify pipette tips between samples and reservoirs betwixt reagents.
j. Occasional positive and negative values close to the cut off value. You should set 3 duplicates for the same sample and the same outcome over 2 samples.
k. Cross contamination when adding samples. You should avoid cross contamination when calculation samples.
Why is abnormal colour developed?
a. Cross contamination during transmission washing. You should reduce the cantankerous contamination by promptly removing the liquid in wells after 3 times of filling washing buffer during manual washing and and so setting the soak time the next times.
b. Cantankerous contamination when patting the plate. Yous should use proper paper towels when patting the plate. Do non bring unrelated materials into the plate. Do not pat at the same place to avoid cross contagion.
c. Contamination due to long storage of samples. You should keep samples fresh or store them under depression temperate to avoid contagion.
d. Abnormal developed color due to bereft filling or as well much residual when the plate washer is obstructed. Yous should fully fill up into every well with pipette but no overflows are allowed. Check that all ports of the plate washer are unobstructed to ensure sufficient washing.
due east. Coagulationor interference of precipitates or residual cell caused by incomplete centrifugation of samples. Yous should complete centrifugation of serum and plasma.
f. Incorrect training of launder buffer or misuse of concentrated launder buffer. Y'all should set wash buffer as manual required.
Besides that, a sweet guy in our company also writes an article about Pre-experiment. If you want to run across the total article, you can click the following link to enter the site: https://www.cusabio.com/1000-226.html.
rowallanwintakephe.blogspot.com
Source: https://www.cusabio.com/c-15091.html
0 Response to "Do You Read an Elisa With the Stop Solution"
Post a Comment